यहाँ क्लास 10वीं साइंस का टेस्ट सीरीज दिया गया है।
class 10th science mcq question
class 10th science mcq question
class 10th science mcq question
Phenomenon – Time (3 min)
Q1
Amount of energy transformation takes place at each trophic level in a food chain is
(A) 1%
(B) 10%
(C) 0.1%
(D) All of energy
Answer – B
Solution a As per 10% law, after every tropic level 10% energy is transferred.
Natural Resources – 3 min
Q2
If a light ray is coming from air to a medium changes velocity to 1.24 x 108 m/s then refractive index of medium is
(A) 1.55
(B) 2.42 : Answer
(C) 2.1
(D) 1.4
Solution à
n21 = V1 (air)/ V2 (medium)
n21 = 3 x 108 / 1.24 x 108
n21 = 2.42
Effects of current – 3 min
Q3.
The length of a wire is halved , but its cross-section remains the same, then resistance becomes
(A) 4 times
(B) 2 times
(C) 1/2 times ( Answer )
(D) 8 times
Solution a Resistance of a wire is directly proportional to the length and inversely proportional to the area of cross-section.
class 10th science mcq question
Natural Phenomenon – 2min
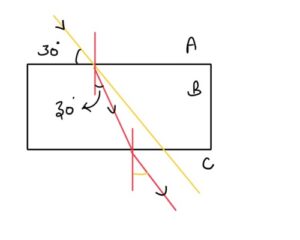
Ques – 4 In given figure light undergoes refraction from A,B and C mediums. Speed of light in medium A is ‘V’ , then find speed of light in medium B also find angle of emergence.
- √2 v , 45°
- √3 v , 30°
- 3v√3 , 60°
- √3v , 60°
3
Answer: D
Solution –
Difficulty Level: Hard
Time to solve: 06 min
Topic: natural phenomenon
Natural Resources – 5min
Q5.
Match the columns
(1) (A) – (s), (B) – (p), (C) – (q), (D) – (r)
(2) (A) – (p), (B) – (s), (C) – (q), (D) – (r)
(3) (A) – (s), (B) – (p), (C) – (r), (D) – (q)
(4) (A) – (s), (B) – (q), (C) – (p), (D) – (r)
Solution – (1)
Natural Phenomenon – 5 min
Q6.
A 4 times big image is formed on screen. Object and screen are at a distance of 120cm , find type of lens used and the focal length of the lens.
- Convex lens , f = 18.4cm
- Convex lens , f = 19.2cm
- Concave lens, f = 18.4cm
- Concave lens, f = 19.2cm
Correct Answer: D
Solution,
m= 4 times {formed by convex lens(Real and Inverted)}
m = – 4
As object and image 120cm apart,
I,e. -u + v = 120cm
v = 120 + u
As, m = v/u
-u = v/u
– u = 120 + u
u
– 4u = 120 + u
– 5u = 120
u = 120/5 = -24cm
Put in V
v = 120 – 24
v = 96cm
Formula for calculation of f – used above
1/f = 1/v – 1/u
1/f = 1/96 – 1/ -24 = 1/96 + 1/24
1/f = 1 + 4 = 5
96 96
f = 19.2cm ….
Difficulty Level: very Hard
Time to solve: 10 min
Topic: natural phenomenon
Effects of Current – 8 mins
Ques – 7 – Calculate the equivalent resistance between A and B –
- 7 ohms
- 1 ohms
- 4 ohms
- 4 ohms
Correct answer: A
Solutions – connecting, R3 , R4 and R5 in parallel combination –
1 = 1 + 1 + 1 = 3
Rx 4 4 4 4
Rx = 4
3
Solving, R6 , R7 in series, Ry = 6 + 6 = 12Ω
Solving, R1 , R2 in series, Rz = 2 + 2 = 04Ω
Solving Ry, Rz,Rq in parallel,
1 = 1 + 1 + 1 = 1+2+2 = 5
Ra 12 6 6 12 12
Ra = 12 Ω
5
solving , Ra, Rx, Rz in series
Rc = 12 + 4 + 4 = 36 + 20 + 60 = 116 = 7.7Ω Ans.
5 3 1 15 15
Difficulty Level: very Hard
Time to solve: 10 mins
Topic: Effects of current.
Q8.
Which of the following are exothermic processes?(i) Combustion
(ii) Nuclear Fission of Uranium-235
(iii) Evaporation of water
(iv) Respiration
(a) (i) and (ii)
(b) (ii) and (iii)
(c) (i) (ii) and (iv): Answer
(d) (ii) and (iv)
Solution
Combustion reaction – An example of a combustion reaction would be the combustion of methane which can be represented as follows: CH4 + O2 → CO2 + H2O + Heat
- The total binding energy releasedin fission of an atomic nucleus varies with the precise break up, but averages about 200 MeV* for U-235
Respiration is the process by which humans take in oxygen and give out carbon dioxide. The chemical equation of this process can be represented as follows:
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy
Q9.
Find the odd man out of the given acids based on there ionization.
- HCL
- HBR
- HF: Answer
- HI
Solution:
Hydrofluoric Acid (HF)
Hydrofluoric acid is a weak acid with the chemical formula HF. This compound is considered to be a weak acid because it does not completely dissociate into hydrogen and fluoride ions when dissolved in water.
Whereas , rest all are strong acids & completely ionised in water and thus produces a large amount of hydrogen ions
HCl (aq) H+ (aq) + Cl– (aq)
The single arrow pointing towards right in the above equation indicates that hydrochloric acid is completely ionised to form ions.
class 10th science mcq question
Q10.
Which of the following metals reacts with water
- Potassium
- Sodium
- Magnesium
- Calcium
- (i) & (ii)
- (ii) & (iii)
- (iii) only
- (i) (ii) & (iv): Answer
Solution
All except Magnesium
- Sodium and potassium react with waterto produce hydrogen gas and the hydroxides of the metals.
- They get softer and more reactive as you move down the period from sodiumto potassium
- Magnesium doesnot react with water to any significant extent. This is in contrast with calcium, immediately below magnesium in the periodic table, which does react slowly with cold
Q11.
Find the correct order of Aerobic Respiration.
- GlucoseàGlycolysis àPyruvate à Kreb’s Cycle àATP : Answer
- Glucoseà Pyruvate à Glycolysis à Kreb’s Cycle àATP
- Glycolysis à Glucose à Kreb’s Cycle à Pyruvate àATP
- Glycolysis à Glucose à Pyruvate à Kreb’s Cycle àATP
class 10th science mcq question
Solution:
The respiration which uses oxygen is called as aerobic respiration.
The process is as follows
Q12.
An element has electronic configuration 2,8, 1 reacts with an element X from group 17.
Explain what will be the chemical formula for the compound that would be formed
- KX
- NaX2
- KX2
- NaX: Correct answer
Solution:
As per given electronic configuration ie. 2,8,1 à Element is Sodium (Na)
Valency of sodium = 1
The group 17 is the Halogen group having valency = 1
Na X
- 1
The compound will be NaX
Q13.
The functions of Testes in human beings is/are-
- To produce hormones like estrogen and progesterone.
- To produce hormones like testosterone.
- To produce the male gamete – sperm.
- To produce the female gamete – ovum.
- I & II
- III Only
- II & IV
- II & III: Answer
Solution:
Testes are the male gonads in human beings. Their functions are
• To produce male hormones like testosterone & the male gamete sperm.
Ovaries are the female gonads in human beings. Their functions are
• To produce female hormones like estrogen and progesterone & the female gamete – ovum.
Q14.
If the genotype (gene combination) is IA IA, then the blood group of the person is A.
if the genotype is IA IO then the blood group is _____
- Blood group A: Answer
- Blood group O
- Blood group B
- Blood group AB
Solution
- Genes IA and IB both are dominant over the gene IO .
- The blood gene IO is recessive in relation to genes IA and IB.
- If the genotype (gene combination) is IA IA, then the blood group of the person is A.
- if the genotype is IA IO even then the blood group is A (because IO is a recessive gene).
Q15.
Which of the following is not a type of Artificial vegetative propagation?
- Cuttings
- Tissue culture
- Tubers: Anwer
- Grafting
Solution:
Tubers are swollen portions of an underground stem that store food so a plant can lie dormant over the winter, for example, potatoes.
It’s a type of Natural vegetative propagation
Whereas,
Artificial vegetative propagation is a type of plant reproduction that involves human intervention.
The most common types of artificial vegetative reproductive techniques include cutting, layering, grafting, suckering, and tissue culturing
class 10th science mcq question part -2 will be updated soon. Stay tuned